2024-02-21
Xeligekimab in moderate to severe plaque psoriasis
Dermatology and Venereology
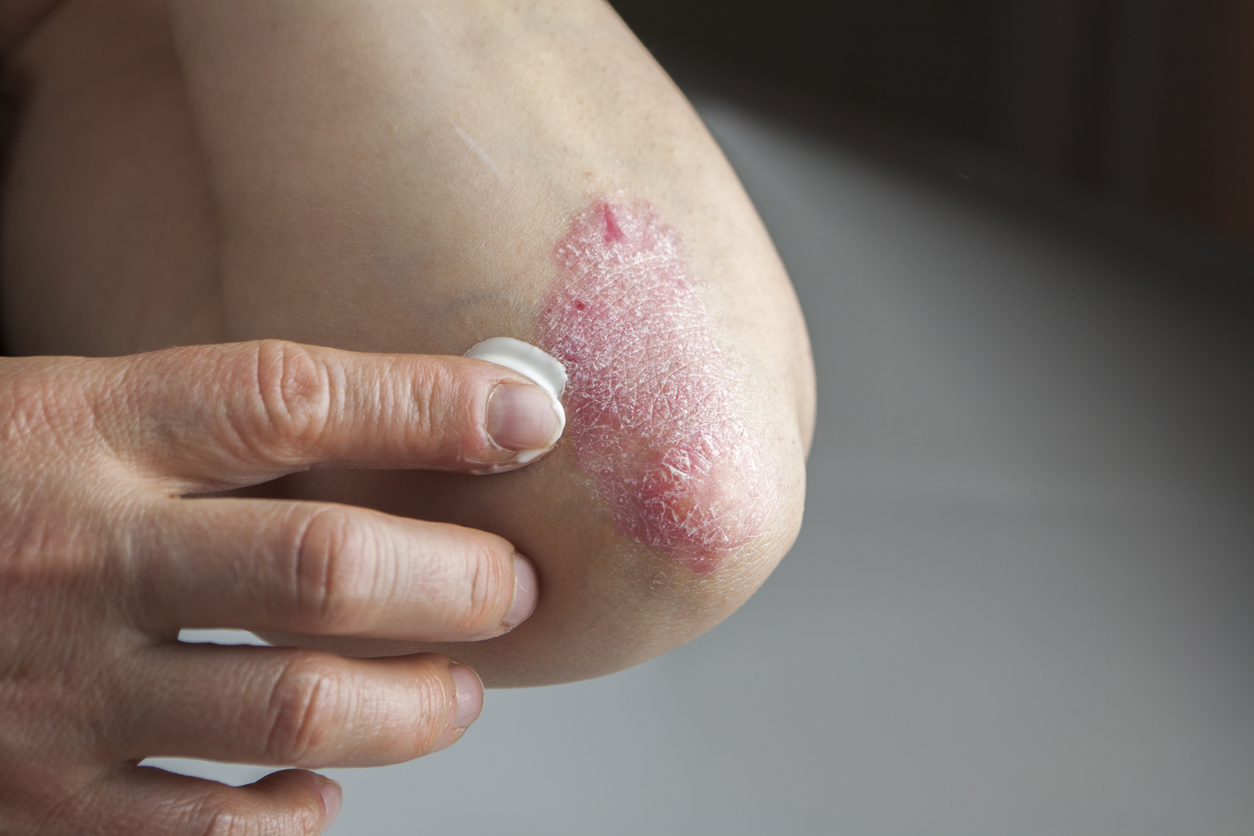
In this study of 420 Chinese patients, researchers evaluated the efficacy and safety of xeligekimab in moderate to severe plaque psoriasis. Xaligekimab is a selective IL-17 inhibitor. Patients were randomized to receive xeligekimab 200 mg every 2 weeks or placebo for an initial period of 12 weeks. Treatment was then continued every 4 weeks for a further 40 weeks. At week 12, PASI scores 75, 90 and 100 were achieved in 90.7%, 74.4% and 30.2% of xeligekimab-treated patients, compared with 8.6%, 1.4% and 0% in the placebo group. No unexpected adverse events were observed.
Last press reviews
What If Female Immunity Should Be Studied Across the Entire Lifespan?

By Ana Espino | Published on June 09, 2026 | 4 min read<br>
GLP-1 Analogues and Polycystic Ovary Syndrome: A New Approach Beyond Weight Loss?

By Ana Espino | Published on June 08, 2026 | 4 min read<br><br><br>P...
Andes Hantavirus: The Next Virus to Watch Closely?

By Ana Espino | Published on June 08, 2026 | 4 min read<br><br><br>